Biosimilars/Research
|
Posted 01/07/2022
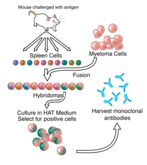
The antitumour necrosis factor-alpha (anti-TNF‑a) monoclonal antibody adalimumab is used to treat a variety of chronic immune-mediated inflammatory diseases, including rheumatoid arthritis, psoriasis, psoriatic arthritis, juvenile arthritis and inflammatory bowel disease. The biosimilar MSB11022 (Idacio/Kromeya) has demonstrated physicochemical and functional similarity to reference adalimumab (Humira) in the preclinical setting. Further study in the clinical setting has established equivalent pharmacokinetics (PK) and efficacy, and comparable safety and immunogenicity for MSB11022 versus adalimumab. MSB11022 is available in three delivery formats: pre-filled syringe, vial and autoinjector. Offering a choice of devices can address patient needs and potentially improve adherence to therapy. Self-injection via pre-filled syringes may be challenging for some patients, due to issues such as needle phobia, pain-related concerns and arthritis hand pain making self-injection more difficult. The availability of alternative self-injection devices allows patients to select a device that suits their needs.