On 29 August 2022, the ranibizumab biosimilar Ranivisio (FYB201), developed by Bioeq – a joint venture between Formycon and Polpharma Biologics, was granted marketing approval by the European Commission (EC).
Ranivisio (FYB201) is to be exclusively commercialized by Teva Pharmaceuticals in Europe and selected other countries through a strategic partnership. This is now the second ophthalmology biosimilar referencing Lucentis to be approved in the European Union (EU) [1].
Ranibizumab is a monoclonal antibody fragment created from the same parent mouse antibody as bevacizumab. Ranibizumab inhibits angiogenesis (the formation of new blood vessels) by inhibiting vascular endothelial growth factor alpha (VEGF-A), a mechanism similar to bevacizumab [2].
The EC approval follows a positive opinion issued in June 2022 by the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) [3] and is applicable to all 27 European Union Member States plus Iceland, Norway and Liechtenstein. The product has already been approved in the UK where it will be marketed under the brand name Ongavia [4].
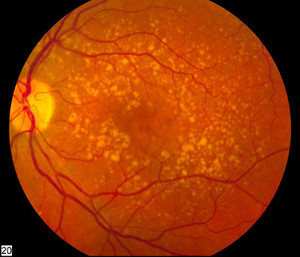
The approval is based on a randomized, double-masked, parallel group, multicentre phase III study, where ranibizumab was shown to be highly similar to its reference medicine in terms of clinical efficacy and ocular and systemic safety in the treatment of age-related macular degeneration (AMD) and its other ophthalmology indications. As such, Ranivisio is now licensed in adults to treat AMD and four other ophthalmology indications: visual impairment due to macular oedema secondary to retinal vein occlusion (branch RVO or central RVO), visual impairment resulting from diabetic macular oedema (DME), proliferative diabetic retinopathy (PDR), and choroidal neovascularisation (CNV). These are the same indications as for the originator product, Lucentis (ranibizumab), which is marketed by Genentech (Roche)/Novartis.
On 2 August 2022, Polpharma Biologics/Formycon/Bioeq announced that they had received approval from the US Food and Drug Administration for their ranibizumab biosimilar (CHS-201/FYB201), Cimerli (ranibizumab-eqrn), the first interchangeable ranibizumab biosimilar approved in the US [5].
Related articles
Canada approves ranibizumab biosimilar Byooviz
Phase III trial evidence used in approval of ranibizumab biosimilar Byooviz
Teva signs deal with Bioeq for ranibizumab biosimilar FYB201
| LATIN AMERICAN FORUM The new section of the ‘Latin American Forum’ on GaBI has been launched. The objective of this new section is to provide you with all the latest news and updates on developments of generic and biosimilar medicines in Latin America in Spanish. View the latest headline article: Panorama regulatorio de los biosimilares en América Latina Browse the news in the Latin American Forum! Register to receive the GaBI Latin American Forum newsletter. Inform colleagues and friends of this new initiative. FORO LATINOAMERICANO Se ha lanzado la nueva sección del ‘Foro Latinoamericano’ sobre GaBI. El objetivo de esta nueva sección es brindarle las últimas noticias y actualizaciones sobre desarrollos de medicamentos genéricos y biosimilares en América Latina en español. Ver el último artículo de cabecera: Panorama regulatorio de los biosimilares en América Latina !Explore las noticias en el Foro Latinoamericano! Regístrese para recibir el boletín informativo GaBI Foro Latinoamericano. Informe a colegas y amigos sobre esta nueva iniciativa.
|
References
1. GaBI Online - Generics and Biosimilars Initiative. EC and FDA approval for first ranibizumab biosimilar Byooviz [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2022 Sep 16]. Available from: www.gabionline.net/biosimilars/news/ec-and-fda-approval-for-first-ranibizumab-biosimilar-byooviz
2. GaBI Online - Generics and Biosimilars Initiative. Biosimilars of ranibizumab [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2022 Sep 16]. Available from: www.gabionline.net/biosimilars/general/Biosimilars-of-ranibizumab
3. GaBI Online - Generics and Biosimilars Initiative. Ranibizumab biosimilar, FYB201, receives EMA recommendation [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2022 Sep 16]. Available from: www.gabionline.net/biosimilars/news/ranibizumab-biosimilar-fyb201-receives-ema-recommendation
4. GaBI Online - Generics and Biosimilars Initiative. Lucentis biosimilars approved in the UK and Korea [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2022 Sep 16]. Available from: www.gabionline.net/biosimilars/news/lucentis-biosimilars-approved-in-the-uk-and-korea
5. GaBI Online - Generics and Biosimilars Initiative. FDA approves first interchangeable ranibizumab biosimilar [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2022 Sep 16]. Available from: www.gabionline.net/biosimilars/news/fda-approves-first-interchangeable-ranibizumab-biosimilar
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Copyright – Unless otherwise stated all contents of this website are © 2022 Pro Pharma Communications International. All Rights Reserved.








 1
1








Post your comment