Dr Elena Wolff-Holz gave a presentation at the European Commission’s Multi-Stakeholder Conference, which was held in Brussels, Belgium on 14 September 2018 giving an overview of biosimilars approved in Europe to date [1].
Dr Wolff-Holz works at the Paul Ehrlich Institut, Federal Agency for Vaccines and Biomedicines and is Chair of the Biosimilar Medicinal Products Working Party (BMWP) of the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP).
Her presentation highlighted how the regulation of biosimilars in the European Union (EU) has evolved since legislation on medicines including biologicals (Directive 2001/83/EC) was passed into law in 2001.
EMA first introduced overarching guidelines for biosimilars in 2005 and approved its first biosimilar Omnitropin (somatropin) in 2006 [2]. Since those initial guidelines were first published the agency has also released many product-class specific guidelines [3].
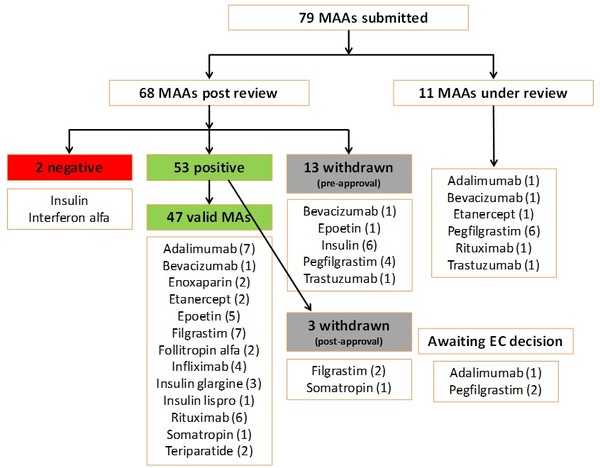
Since EMA started reviewing applications for biosimilars, and up to September 2018, the agency had received 79 marketing authorization applications (MAAs). Of these, 11 were still under review and 13 had been withdrawn before approval was granted (six for insulin biosimilars, one for bevacizumab biosimilar, one for epoetin biosimilar, four for pegfilgrastim biosimilars and one for trastuzumab biosimilar). Up to September 2018, the agency had given a positive opinion for 53 MAAs [of which three were later witdrawn (two for filgrastim and one for somatropin)] and a negative opinion for two MAAs (interferon alfa and insulin). There were also one adalimumab biosimilar and two pegfilgrastim biosimilars awaiting a final decision by the European Commission (EC), see Figure 1.
Figure 1: Biosimilars reviewed by EMA (to September 2018)
EC: European Commission; EMA: European Medicines Agency; MAAs: marketing authorization applications.
In September 2018, the adalimumab biosimilar Hulio (FKB327) and the pegfilgrastim biosimilar Pelgraz received approval in the EU. In November 2018, the pegfilgrastim biosimilars Fulphila (MYL 1401H), Pelmeg (B12019) and Ziextenzo (LA-EP2006) also received approval in the EU. In December 2018, the trastuzumab biosimilar Ogivri (MYL-1401O) received approval in the EU [4].
In December 2018, the EMA’s CHMP gave a positive opinion for the bevacizumab biosimilar, Zirabev (PF 06439535). And in January 2019, the CHMP also gave positive opinions for the adalimumab biosimilars, Idacio and Kromeya (MSB11022) [4].
Conflict of interest
The author of the presentation [1] stated that the views presented in the presentation were her own and do not necessarily reflect the views of the Paul-Ehrlich-Institut or EMA.
Related article
Biosimilar clinical trials as confirmatory evidence
References
1. Wolff-Holz E. Biosimilars use in oncology: overview by the European Medicines Agency on biosimilars approved so far. EC Multi-Stakeholder Conference; 14 September 2018; Brussels, Belgium.
2. GaBI Online - Generics and Biosimilars Initiative. Biosimilars use in Europe [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2019 Apr 1]. Available from: www.gabionline.net/Reports/Biosimilars-use-in-Europe
3. GaBI Online - Generics and Biosimilars Initiative. EU guidelines for biosimilars [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2019 Apr 1]. Available from: www.gabionline.net/Guidelines/EU-guidelines-for-biosimilars
4. GaBI Online - Generics and Biosimilars Initiative. Biosimilars approved in Europe [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2019 Apr 1]. Available from: www.gabionline.net/Biosimilars/General/Biosimilars-approved-in-Europe
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Copyright – Unless otherwise stated all contents of this website are © 2019 Pro Pharma Communications International. All Rights Reserved.








 0
0










Post your comment