Singapore-based Prestige Biopharma (Prestige) has formed an agreement with Russian company Pharmapark to commercialize Prestige’s bevacizumab non-originator biological in Russia. Russian company Geropharm has also launched an insulin aspart non-originator biological on the market in the country.
The agreement between Prestige and Pharmapark was announced on 15 April 2021 and represents an exclusive partnership for the commercialization of Prestige’s bevacizumab non-originator biological, HD204, in Russia.
Bevacizumab is a humanized monoclonal antibody. It inhibits angiogenesis (the formation of new blood vessels) by blocking the action of vascular endothelial growth factor A (VEGF-A). Bevacizumab can therefore slow the growth of new blood vessels in tumours and is used to treat various cancers, including colorectal, lung, breast, glioblastoma, kidney and ovarian.
HD204 is currently in phase III clinical trials, with patient recruitment ongoing. Phase I trials on the pharmacokinetics, safety and immunogenicity of HD204 compared to the originator product reported positive results. Under the agreement, Pharmapark will have exclusive rights to commercialize HD204 in Russia. Prestige will have responsibility for supply of the product from its manufacturing site in Osong, South Korea, although Pharmapark will have the option to manufacture the product in Russia.
This is not the first collaboration between the two companies. In 2019, Prestige formed a deal with Pharmapark for the supply of Prestige’s trastuzumab biosimilar, HD201, in Russia [1]. HD201 was accepted for review by the European Medicines Agency in May 2019 [2] and is to be distributed in Europe by UK-based firm Mundipharma [3].
Of the recent agreement, CEO of Prestige Lisa S Park commented: ‘We are very pleased to further extend our partnership with Pharmapark to commercialize our second biosimilar program in the Russian Federation. We are excited to bring another high-quality product to this key market with the ultimate goal to make the product available globally’.
Vyacheslav Lebedyansky, CEO of Pharmapark, added: ‘This extension to our existing partnership with Prestige BioPharma is another important milestone for Pharmapark allowing our company to bring vital and affordable medicines to Russian population in line with the Russian Pharma-2030 strategy. Bevacizumab biosimilar candidate will be an excellent addition to our growing biosimilar portfolio’.
In other news for the Russian biologicals market, Geropharm, which is headquartered in St Petersburg, recently launched their non-originator insulin aspart in the country.
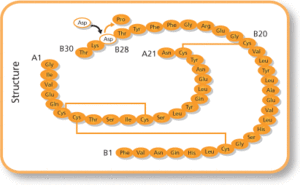
At the end of 2020, Geropharm received registration for their insulin aspart non-originator biological, which has the brand name RinFast. The product will be sold in cartridges and pre-filled, multi-dose syringe pens. Rinfast is a copy of Novo Nordisk’s product, which was previously the only insulin aspart available in Russia.
The approval follows pharmacological, preclinical and clinical studies, which were partially supported by the Russian Ministry of Industry and Trade under the Pharmamed-2020 program. Sergey Tsyb, First Deputy Minister of Industry and Trade of the Russian Federation, said: ‘The Pharmamed-2020 program gave a powerful impetus to the domestic pharmaceutical industry and actually helped to revive it. Today, manufacturers have accumulated enough competencies to further develop imported-independent medicines in the Russian Federation […]’.
‘For Geropharm, Russia is the home region, and we are focused on comprehensive solution of problems within the framework of the direction of diabetes here. This is the development and production of effective and safe medicines, as well as active participation in the social agenda […],’ said Peter Rodionov, CEO of Geropharm. ‘In the coming years, we will continue to develop a portfolio of insulins and increase the availability of analogues for Russian patients […],’ he added.
Related articles
EC approval for insulin aspart biosimilar Kixelle
EMA recommends approval of four bevacizumab biosimilars
Positive phase III results for trastuzumab biosimilar HD201
| LATIN AMERICAN FORUM – Coming soon! To further enhance the objectives of GaBI in sharing information and knowledge that ensure policies supportive of safe biosimilars use, we are pleased to announce that we will be launching a new section on GaBI Online and GaBI Journal, the ‘Latin American Forum’ (in Spanish) featuring the latest news and updates on research and developments in generic and biosimilar medicines in Latin America. Register to receive the GaBI Latin American Forum newsletter. Inform colleagues and friends of this new initiative. LATIN AMERICAN FORUM – Próximamente! Para fomentar los objetivos de GaBI sobre la difusión de información y conocimiento sobre las políticas de apoyo que garantizan el uso seguro de medicamentos biosimilares, nos complace anunciar el lanzamiento de una nueva sección en GaBI Online y GaBI Journal, el ‘Latin American Forum’ (en español), que presentará las últimas noticias y actualizaciones en investigación y desarrollo sobre medicamentos genéricos y biosimilares en Latinoamérica. Regístrese para recibir el boletín informativo GaBI Latin American Forum. Informe a colegas y amigos sobre esta nueva iniciativa.
|
References
1. GaBI Online - Generics and Biosimilars Initiative. Prestige and Pharmapark make deal for trastuzumab biosimilar [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2021 May 14]. Available from: www.gabionline.net/Pharma-News/Prestige-and-Pharmapark-make-deal-for-trastuzumab-biosimilar
2. GaBI Online - Generics and Biosimilars Initiative. Trastuzumab biosimilar from Prestige accepted for review by EMA [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2021 May 14]. Available from: www.gabionline.net/Biosimilars/News/Trastuzumab-biosimilar-from-Prestige-accepted-for-review-by-EMA
3. GaBI Online - Generics and Biosimilars Initiative. Trastuzumab biosimilar to be distributed by Mundipharma [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2021 May 14]. Available from: www.gabionline.net/Biosimilars/News/Trastuzumab-biosimilar-to-be-distributed-by-Mundipharma
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Copyright – Unless otherwise stated all contents of this website are © 2021 Pro Pharma Communications International. All Rights Reserved.
Source: Geropharm; Prestige Biopharma








 0
0









Post your comment