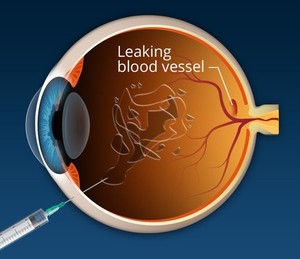
In March 2021, Formycon confirmed the resubmission strategy for their Lucentis® (ranibizumab) biosimilar candidate (FYB2010) in the US. In addition, Alteogen announced the completion of its phase I clinical trial of Eylea® (aflibercept) biosimilar (ALT-L9). Both of these biological drugs are used to treat conditions leading to the loss of vision.
Formicon’s Lucentis biosimilar
Formycon and its licensing partner Bioeq AG engaged in successful pre-Biologics License Application (BLA) interactions with the US Food and Drug Administration (FDA). This led to the November 2020 announcement that the initial submission strategy for FYB201 was to be adjusted as part of a simplification of the approval procedure.
Now, with the optimization of the commercial supply chain, FYB201’s approval will occur directly at commercial scale. The BLA-submission is expected to be filed during the first half of 2021, with submission to the European Medicines Agency (EMA) and other highly regulated territories such as Canada, Australia, Switzerland and the UK to follow in due course.
Ranibizumab inhibits vascular endothelial growth factor (VEGF) which is responsible for the excessive formation of blood vessels in the retina leading to progressive loss of vision. The patents on Lucentis have already expired in the US and is due to expire in Europe in 2022 [1].
Alteogen’s Eylea biosimilar
Alteogen conducted its phase I clinical trial of ALT-L9 at four major hospitals in South Korea. The randomized, double-blinded, active control and parallel trial was carried out in 28 patients with neovascular (wet) age-related macular degeneration. It confirmed the safety and efficacy of ALT-L9 were similar to that of Eylea. Alteogen will now prove similarity of its product with Eylea through a phase III clinical trial.
Like ranibizumab, aflibercept is also a VEGF inhibitor. It is indicated for the treatment of neovascular (wet) age-related macular degeneration, macular edema following retinal vein occlusion, diabetic macular edema and diabetic retinopathy in patients with diabetic macular edema [2].
Eylea is Regeneron’s blockbuster drug with a reported market size of US$4.947 billion in 2020 in the US alone. Alteogen now has a strong position in the market as it has obtained a formulation patent registered in European Union, Japan and the US and a process patent for its method of producing the aflibercept biosimilar [3].
Related articles
FDA accepts application for ranibizumab biosimilar
Japanese firms collaborate on Eylea biosimilar
| LATIN AMERICAN FORUM – Coming soon! To further enhance the objectives of GaBI in sharing information and knowledge that ensure policies supportive of safe biosimilars use, we are pleased to announce that we will be launching a new section on GaBI Online and GaBI Journal, the ‘Latin American Forum’ (in Spanish) featuring the latest news and updates on research and developments in generic and biosimilar medicines in Latin America. Register to receive the GaBI Latin American Forum newsletter. Inform colleagues and friends of this new initiative. LATIN AMERICAN FORUM – Próximamente! Para fomentar los objetivos de GaBI sobre la difusión de información y conocimiento sobre las políticas de apoyo que garantizan el uso seguro de medicamentos biosimilares, nos complace anunciar el lanzamiento de una nueva sección en GaBI Online y GaBI Journal, el ‘Latin American Forum’ (en español), que presentará las últimas noticias y actualizaciones en investigación y desarrollo sobre medicamentos genéricos y biosimilares en Latinoamérica. Regístrese para recibir el boletín informativo GaBI Latin American Forum. Informe a colegas y amigos sobre esta nueva iniciativa.
|
References
1. GaBI Online - Generics and Biosimilars Initiative. Biosimilars of ranibizumab [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2021 Apr 16]. Available from: www.gabionline.net/Biosimilars/General/Biosimilars-of-ranibizumab
2. GaBI Online - Generics and Biosimilars Initiative. Biosimilars of aflibercept [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2021 Apr 16]. Available from: www.gabionline.net/Biosimilars/General/Biosimilars-of-aflibercept
3. GaBI Online - Generics and Biosimilars Initiative. Alteogen wins process patent for aflibercept biosimilar [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2021 Apr 16]. Available from: www.gabionline.net/Biosimilars/General/Alteogen-wins-process-patent-for-aflibercept-biosimilar
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Copyright – Unless otherwise stated all contents of this website are © 2021 Pro Pharma Communications International. All Rights Reserved.








 0
0








Post your comment