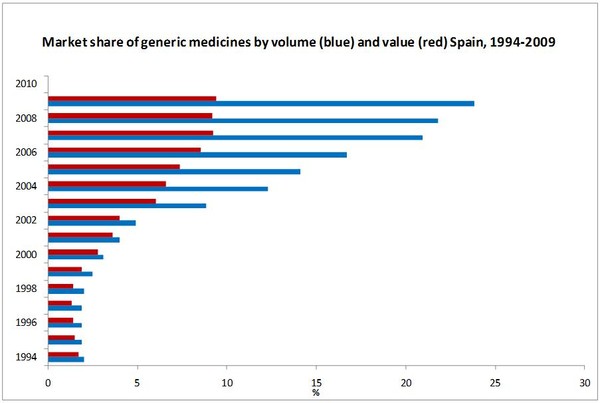
The market share of generic medicines was small in the second half of the 1990s, hovering around 1.5% in terms of value and 2% in terms of volume. The 2000 and 2003 generic medicine policies appear to have had a limited impact on the development of the Spanish generic medicines market [1].
With respect to value of consumption, market shares of generic medicines have grown from 1.7% in 1994 to 9.4% in 2010. With respect to volume of consumption, market shares of generic medicines increased from 2.0% in 1994 to 24% in 2010 [1, 2].
Source: 1994–2002 [1], 2003–2009 [2].
Spain has pricing regulation of generic medicines, which means that penetration of generic medicines is less successful than in countries that permit (relatively) free pricing of medicines, e.g. Germany, The Netherlands, UK [3].
The legacy of counterfeit products, along with reference pricing, which erodes price differentials between off-patent brands and their generic versions, as well as the few incentives for physicians to prescribe generics or for pharmacists to dispense cheaper alternatives, undermine the market. Because of these factors, generics consumption in Spain remains among the lowest in the EU, although the Spanish market is characterised by high use of biologicals and a rapid uptake of new products [4].
Physicians are not obliged or stimulated to prescribe by international non-proprietary name (INN) [1].
Patient co-payments are slightly lower for generics [1].
The Spanish government has launched several advertising campaigns to inform patients of generic medicines [5].
Highlights of the generics market in Spain
- Market share of generic medicines by volume is 24%, accounting for 9.4% in value [2].
- Historically, the Spanish generics market has been small due to the existence of a developed market of copies and low medicine prices [1].
- Competition created by the 2000 generic medicines policy benefited pharmacists rather than patients, thus inhibiting demand for generics [1].
- The 2003 generics medicines policy incited companies to reduce prices of originator medicines to reference prices that are set close to marginal costs, thus limiting the profitability of and demand for generics [1].
- Few incentives exist for physicians to prescribe generics [1].
- Pharmacists are financially penalised for dispensing generics [1].
- A recent amendment to the law includes all biotechnology medicines on a list of products that cannot be automatically substituted [1].
References
1. Simoens S, De Coster S. Sustaining Generic Medicines Markets in Europe. April 2006. [monograph on the Internet]. Brussels, Belgium, European Generic medicines Association (EGA) [cited 2011 October 14]. Available from: www.egagenerics.com/doc/simoens-report_2006-04.pdf
2. Ministerio de Sanidad Politica Social e Igualdad [Ministry of Health Social Policy and Equality] . Consumo de genéricos en España [Spanish] [Consumption of Generic Drugs in Spain] [page on the Internet]. Madrid, Spain [cited 2011 October 14]. Available from: www.medicamentosgenericosefg.es/consumo-genericos-espana.html
3. Simoens S. Generic medicine pricing in Europe: current issues and future perspective. J Med Econ. 2008;11(1):171-5.
4. Pisani J, Bonduelle Y. Opportunities and barriers in the biosimilar market: Evolution or revolution for generics companies? 2006 PricewaterhouseCoopers LLP.
5. Vogler S, Espin J, Habl C. Pharmaceutical Pricing and Reimbursement Information (PPRI) – New PPRI analysis including Spain. Pharmaceuticals Policy and Law. 2009;11(3):213-34.