As comparative clinical studies can be costly and time consuming, the US Food and Drug Administration (FDA), is currently conducting research to inform the agency’s thinking on critical aspects of the use of pharmacodynamic (PD) biomarkers to demonstrate biosimilarity. This, according to the agency could either streamline or negate the need for comparative clinical studies.
A presentation entitled ‘An update on the FDA biosimilar program: progress and directions, 2021’ was given by Dr Peter Stein, Director of the Office of New Drugs at the Center for Drug Evaluation and Research (CDER) of FDA, at the DIA Biosimilars Conference. In his presentation, Dr Stein outlined the ‘positioning’ of PD biomarkers in the evaluation of biosimilarity [1].
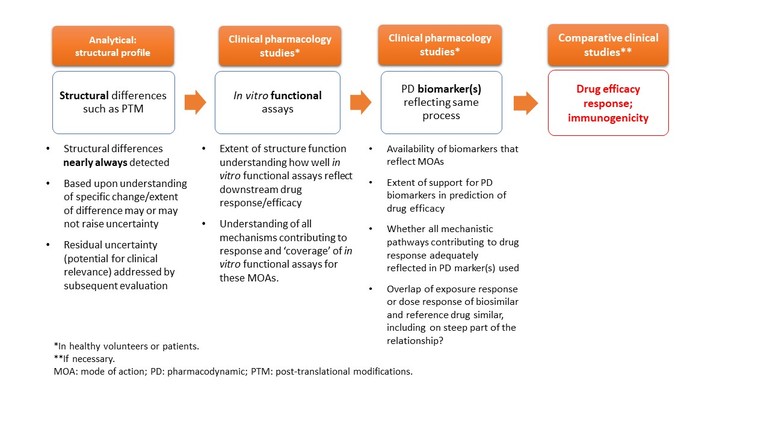
According to FDA’s stepwise approach to demonstrating biosimilarity, the process should begin with the establishment of an analytically ‘highly similar’ foundation from comparative analytical studies (product quality), followed by animal or in vitro studies and clinical studies assessing safety/immunogenicity, pharmacokinetic (PK) similarity and, when appropriate, PD similarity. Additional comparative clinical studies may be necessary to address residual uncertainties that remain to support a demonstration that there are no clinically meaningful differences between the proposed biosimilar and reference product in terms of the safety, purity and potency of the product.
Dr Stein outlined how PD biomarkers fit into this stepwise approach in Table 1.
Table 1: PD biomarkers in the evaluation of biosimilarity
Conflict of interest
The author of the presentation [1] did not provide any conflict-of-interest statement.
Related articles
Key considerations for PD biomarkers in evaluating biosimilarity
The role of PD biomarkers in biosimilarity
PD biomarkers for biosimilar development and approval
| LATIN AMERICAN FORUM The new section of the ‘Latin American Forum’ on GaBI has been launched. The objective of this new section is to provide you with all the latest news and updates on developments of generic and biosimilar medicines in Latin America in Spanish. View last week’s headline article: La evidencia sobre la sustitución automática de biológicos es limitada Browse the news in the Latin American Forum! Register to receive the GaBI Latin American Forum newsletter. Inform colleagues and friends of this new initiative. LATIN AMERICAN FORUM Se ha lanzado la nueva sección del ‘Foro Latinoamericano’ sobre GaBI. El objetivo de esta nueva sección es brindarle las últimas noticias y actualizaciones sobre desarrollos de medicamentos genéricos y biosimilares en América Latina en español. Vea el artículo principal de la semana pasada: La evidencia sobre la sustitución automática de biológicos es limitada Explore las noticias en el Foro Latinoamericano! Regístrese para recibir el boletín informativo GaBI Foro Latinoamericano. Informe a colegas y amigos sobre esta nueva iniciativa.
|
Reference
1. An update on the FDA biosimilar program: progress and directions, 2021. DIA Biosimilars, Virtual Conference, 5-6 October 2021.
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Copyright – Unless otherwise stated all contents of this website are © 2021 Pro Pharma Communications International. All Rights Reserved.








 0
0











Post your comment