On 30 March 2026, the US Food and Drug Administration (FDA) approved Teva Pharmaceuticals’ Ponlimsi (denosumab-adet), a biosimilar to Amgen’s Prolia and Xgeva (denosumab) [1].
FDA approves denosumab biosimilar Ponlimsi
Biosimilars/News
|
Posted 24/04/2026
 0
Post your comment
0
Post your comment
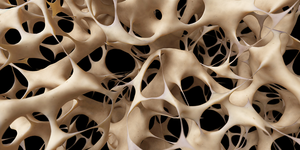
Denosumab is a human monoclonal antibody that binds to RANKL (receptor activator of nuclear factor kappa-Β ligand), a protein essential for the formation, function, and survival of osteoclasts. By inhibiting RANKL, denosumab reduces bone resorption. It is used to treat osteoporosis and bone loss due to cancer, as well as to prevent skeletal-related events in patients with bone metastases [1].
The approval was supported by a totality of evidence, including analytical and clinical data demonstrating similar efficacy, safety, and immunogenicity profiles between Ponlimsi (denosumab-adet) and the reference product, Prolia.
Ponlimsi (denosumab-sldn) is approved for all indications of the reference product, including:
- treatment of postmenopausal women with osteoporosis at high risk for fracture
- treatment to increase bone mass in men with osteoporosis at high risk for fracture
- treatment of glucocorticoid-induced osteoporosis in men and women at high risk for fracture
- treatment to increase bone mass in men at high risk for fracture receiving androgen deprivation therapy for nonmetastatic prostate cancer
- treatment increase bone mass in women at high risk for fracture receiving adjuvant aromatase inhibitor therapy for breast cancer.
The product is supplied as a 60 mg/mL single-use prefilled syringe for subcutaneous injection.
With this approval, Ponlimsi becomes the latest denosumab biosimilar in Teva’s growing biosimilars portfolio. Teva has announced that a commercial launch is expected in the third quarter of 2026.
By end of December 2025, the FDA had approved a total of 18 denosumab biosimilars [2]. There were no other biosimilars approved by the FDA in March 2026, and there were no biosimilars approved in February 2026.
Related articles
FDA approves denosumab biosimilars Osvyrti and Jubereq, Boncresa and Oziltus
ANVISA approves biosimilars for denosumab, trastuzumab, and aflibercept
FDA approves six denosumab biosimilars
|
LATIN AMERICAN FORUM View the latest headline article: La jeringa precargada del biosimilar de aflibercept (AVT06) promete inyecciones oculares más seguras y rápidas Browse the news in the Latin American Forum! Register to receive the GaBI Latin American Forum newsletter. Inform colleagues and friends of this new initiative.
FORO LATINOAMERICANO Ver el último artículo de cabecera: La jeringa precargada del biosimilar de aflibercept (AVT06) promete inyecciones oculares más seguras y rápidas !Explore las noticias en el Foro Latinoamericano! Regístrese para recibir el boletín informativo GaBI Foro Latinoamericano. Informe a colegas y amigos sobre esta nueva iniciativa. |
References
1. GaBI Online - Generics and Biosimilars Initiative. Biosimilars of denosumab [www.gabionline.net].Mol, Belgium: Pro Pharma Communications International; [cited 2026 Apr 24]. Available from:
www.gabionline.net/biosimilars/general/Biosimilars-of-denosumab
2. GaBI Online - Generics and Biosimilars Initiative. Biosimilars approved in the US [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2026 Apr 24]. Available from: www.gabionline.net/biosimilars/general/biosimilars-approved-in-the-us
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Copyright – Unless otherwise stated all contents of this website are © 2026 Pro Pharma Communications International. All Rights Reserved.
Research
Biosimilar aflibercept (AVT06) pre-filled syringe promises safer, faster eye injections
OECD study finds no direct link between advertising rules and biosimilar uptake
General
SBR issues position statement on safe originator biological/biosimilar interchangeability
Samsung Bioepis wins Pyzchiva case; Regeneron patent rulings threaten foreign biosimilars
EMA recommends approval of three new biosimilars for diabetes and autoimmune conditions
Biosimilars/News Posted 16/04/2026
EMA recommends approval for pertuzumab and tocilizumab biosimilars

Biosimilars/News Posted 08/04/2026
The best selling biotechnology drugs of 2008: the next biosimilars targets






Post your comment