STADA and Xbrane Biopharma have announced that the UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has granted a marketing authorization for Ximluci, a biosimilar referencing ophthalmology drug, Lucentis (ranibizumab). In addition, Samsung Bioepis and Samil Pharmaceuticals will launch their biosimilar of Lucentis, Amelivu, in South Korea.
Ranibizumab biosimilar Ximluci and Amelivu to launch in the UK and South Korea
Biosimilars/News
|
Posted 24/02/2023
 0
Post your comment
0
Post your comment
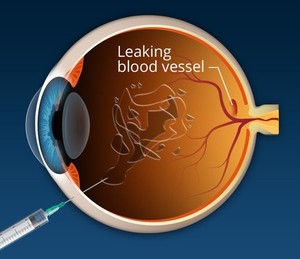
Ranibizumab is a monoclonal antibody fragment created from the same parent mouse antibody as bevacizumab. It inhibits angiogenesis (the formation of new blood vessels) by inhibiting vascular endothelial growth factor A (VEGF-A). Ranibizumab can be used to treat macular degeneration by inhibiting VEGF, which is responsible for the excessive formation of blood vessels in the retina leading to progressive loss of vision. The monoclonal antibody drug is indicated for the treatment of wet age-related macular degeneration (AMD), macular oedema, degenerative myopia and diabetes complications; all conditions of the eye causing vision loss [1].
Ximluci UK authorization
The European Commission granted marketing authorization Ximluci, a biosimilar referencing Lucentis ranibizumab, in 2022 [2]. Both this and the UK authorization are for Ximluci 10 mg/mL solution for injection and the product will launch in the regions in 2023.
Stada and Xbrane entered into an agreement in 2018 under which the two companies are jointly responsible for development and for manufacturing Ximluci. Stada now holds the marketing authorizations and the commercial rights to the biosimilar across all territories included in the agreement, which covers Europe, the US, several countries in the Middle East and North Africa region, and selected Asia-Pacific markets.
Amelivu launch in South Korea
Samsung Bioepis’ ophthalmology biosimilar was approved in the US in September 2021 [3, 4], Europe in August 2021 [5, 6], Canada [7] and South Korea [8]. In the US, it is marketed as Byooviz (ranibizumab-nuna) and is approved for the treatment of neovascular AMD (nAMD), macular oedema following retinal vein occlusion (RVO), and myopic choroidal neovascularization (mCNV) [4]. However, delays to product launch in other regions occurred due to a patent-related agreement between Samsung Bioepis and Lucentis producer Genentech.
Now, Samsung Bioepis’ South Korean launch of biosimilar ranibizumab Amelivu, with partner Samil Pharmaceutical, has finally been announced following the expiration of Genentech’s Lucentis supplementary protection certificate [9].
Related articles
Phase III trial evidence used in approval of ranibizumab biosimilar Byooviz
Canada approves ranibizumab biosimilar Byooviz
Fresh partnerships announced for ranibizumab and trastuzumab biosimilars
Biosimilars approved in South Korea
|
LATIN AMERICAN FORUM View the latest headline article: Directrices revisadas de la OMS para productos biosimilares seguros y eficaces Browse the news in the Latin American Forum! Register to receive the GaBI Latin American Forum newsletter. Inform colleagues and friends of this new initiative.
FORO LATINOAMERICANO Ver el último artículo de cabecera: Directrices revisadas de la OMS para productos biosimilares seguros y eficaces !Explore las noticias en el Foro Latinoamericano! Regístrese para recibir el boletín informativo GaBI Foro Latinoamericano. Informe a colegas y amigos sobre esta nueva iniciativa. |
References
1. GaBI Online - Generics and Biosimilars Initiative. Biosimilars of ranibizumab [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Feb 24]. Available from:
www.gabionline.net/biosimilars/general/Biosimilars-of-ranibizumab
2. GaBI Online - Generics and Biosimilars Initiative. Australia and EU: Alvotech Humira and Stada Lucentis biosimilars approved [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Feb 24]. Available from:
www.gabionline.net/biosimilars/news/australia-and-eu-alvotech-humira-and-stada-lucentis-biosimilars-approved
3. GaBI Online - Generics and Biosimilars Initiative. Biosimilars approved in the US [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Feb 24]. Available from:
www.gabionline.net/biosimilars/general/Biosimilars-approved-in-the-US
4. GaBI Online - Generics and Biosimilars Initiative. Phase III trial evidence used in approval of ranibizumab biosimilar Byooviz [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Feb 24]. Available from:
www.gabionline.net/biosimilars/research/phase-III-trial-evidence-used-in-approval-of-ranibizumab-biosimilar-byooviz
5. GaBI Online - Generics and Biosimilars Initiative. Biosimilars approved in Europe [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Feb 24]. Available from:
www.gabionline.net/biosimilars/general/biosimilars-approved-in-europe
6. GaBI Online - Generics and Biosimilars Initiative. EMA recommends approval of Samsung Bioepis Biogen’s ranibizumab biosimilar Byooviz [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Feb 24]. Available from:
www.gabionline.net/biosimilars/news/ema-recommends-approval-of-samsung-bioepis-biogen-s-ranibizumab-biosimilar-byooviz
7. GaBI Online - Generics and Biosimilars Initiative. Canada approves ranibizumab biosimilar Byooviz [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Feb 24]. Available from:
www.gabionline.net/biosimilars/news/canada-approves-ranibizumab-biosimilar-byooviz
8. GaBI Online - Generics and Biosimilars Initiative. Lucentis biosimilars approved in the UK and Korea [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Feb 24]. Available from:
www.gabionline.net/biosimilars/news/lucentis-biosimilars-approved-in-the-uk-and-korea
9. GaBI Online - Generics and Biosimilars Initiative. New partnerships: Prestige-Intas and Samsung Bioepis-Samil [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2023 Feb 24]. Available from:
www.gabionline.net/pharma-news/new-partnerships-prestige-intas-and-samsung-bioepis-samil
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Copyright – Unless otherwise stated all contents of this website are © 2023 Pro Pharma Communications International. All Rights Reserved.
Research
Reaching ESG goals in pharmaceutical development
What is the future for the US biosimilar interchangeability designation
General
Samsung Bioepis wins Pyzchiva case; Regeneron patent rulings threaten foreign biosimilars
Chinese biosimilars go global: growth, partnerships, and challenges
Most viewed articles
The best selling biotechnology drugs of 2008: the next biosimilars targets
Global biosimilars guideline development – EGA’s perspective
Related content
FDA approves third interchangeable ranibizumab biosimilar Nufymco
FDA approves Poherdy (first interchangeable pertuzumab) and Armlupeg (pegfilgrastim) biosimilars
EMA recommends approval for insulin glargine biosimilar Ondibta and denosumab biosimilar Osqay
FDA approves denosumab biosimilars Osvyrti and Jubereq, Boncresa and Oziltus
FDA approves third interchangeable ranibizumab biosimilar Nufymco

Biosimilars/News Posted 09/02/2026
FDA approves Poherdy (first interchangeable pertuzumab) and Armlupeg (pegfilgrastim) biosimilars

Biosimilars/News Posted 27/01/2026
EMA recommends approval for insulin glargine biosimilar Ondibta and denosumab biosimilar Osqay

Biosimilars/News Posted 16/01/2026
FDA approves denosumab biosimilars Osvyrti and Jubereq, Boncresa and Oziltus

Biosimilars/News Posted 07/01/2026
The best selling biotechnology drugs of 2008: the next biosimilars targets






Post your comment