Biosimilars/General
Approved biosimilars for South Korean firms: Celltrion and Samsung Bioepis
Korean companies, such as Celltrion Healthcare (Celltrion) and Samsung Bioepis (Samsung and Biogen’s joint venture), are becoming more and more important both in the country and worldwide when it comes to developing biosimilars.
Canadian provinces expand their use of biosimilars
New Brunswick has become the third Canadian province to implement a biosimilar switching policy. The province of Alberta has also expanded its switching programme to include biosimilars of the anti-inflammatory treatment adalimumab (Humira).
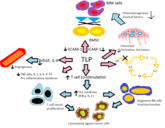
Safety of switching between rituximab biosimilars in cancer
In more than 10 years of clinical experience, no substantial clinical and safety differences have been detected among biosimilars and their already approved biologicals [1]. However, concerns are raised with respect to the practice of switching in patients already treated with a specific biological product (either reference or biosimilar) [2].
Insurer formularies increase complexity for healthcare providers
An assessment of insurer formularies for pegfilgrastim in the US suggests that payer-imposed preferences increase complexity when pharmacists and physicians are choosing which version of a drug to stock and administer.
Rheumatoid arthritis treatments in Australia
Analysis of the treatment landscape for rheumatoid arthritis in Australia shows that AbbVie’s Humira (adalimumab) and Pfizer’s Enbrel (etanercept) together make up half of total sales.
Biosimilars of enoxaparin sodium
Enoxaparin sodium is an anticoagulant medication (blood thinner). It is used to treat and prevent deep vein thrombosis (DVT) and pulmonary embolism (PE) including during pregnancy and following certain types of surgery. It is also used in those with acute coronary syndrome (ACS) and heart attacks.
British Columbia adds adalimumab to biosimilar switching programme
British Columbia (BC), the first province in Canada to switch patients to biosimilar drugs, has added adalimumab to its switching programme. Almost 6,000 patients will be transitioned from originator adalimumab (Humira) to one of five biosimilar versions.
Biosimilars of ustekinumab
Ustekinumab is a humanized monoclonal antibody that interferes with the triggering of the body’s inflammatory response through the suppression of certain cytokines. Specifically, it blocks interleukin IL 12 and IL 23 which help activate certain T cells. It binds to the p 40 subunit of both IL 12 and IL 23 so that they subsequently cannot bind to their receptors. It is indicated for the treatment of Crohn’s disease, psoriasis and psoriatic arthritis.
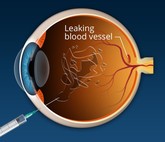
Biosimilars of aflibercept
Aflibercept is a vascular endothelial growth factor (VEGF) inhibitor. It is indicated for the treatment of neovascular (wet) age-related macular degeneration, macular oedema following retinal vein occlusion, diabetic macular oedema and diabetic retinopathy in patients with diabetic macular oedema.
Copy biologicals approved in China
Last update: 2 April 2021
In China, the regulatory body for the approval of medicines, including biologicals, is the National Medical Products Administration (NMPA), formerly the China Food and Drug Administration (CFDA: simplified Chinese: 国家食品药品监督管理局).