Biosimilars
FDA approves pegfilgrastim and rituximab biosimilars
The US Food and Drug Administration (FDA) has approved the pegfilgrastim biosimilar Fylnetra (pegfilgrastim-pbbk) and the rituximab biosimilar Riabni (rituximab-arrx).
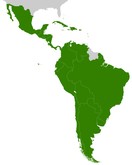
Current status of monoclonal antibody biosimilars approved in Latin America
By the end of 2021, biosimilar antibodies of rituximab, trastuzumab, infliximab, adalimumab and bevacizumab were expected to be commercialized in Latin America with 25 different brand names.
Byooviz: first ophthalmology biosimilar launches in US
Biogen and Samsung Bioepis have launched Byooviz (ranibizumab-nuna) on the US market as the first ophthalmology biosimilar. It references Roche’s (Genentech’s) blockbuster therapy, Lucentis.
The impact of biosimilar insulins on public spending in Brazil
The availability of insulin for the treatment of diabetes mellitus, a chronic disease that increases with age, and its financial sustainability are of general concerns, especially for universal healthcare systems, such as the Brazilian Unified Health System (Sistema Único de Saúde, SUS) [1]. This is why similar biotherapeutic products (produtos bioterapêuticos similares, SBP) can be a cost reduction strategy in improving access to essential supplies for the population. These drugs demand less investment and acquisition cost [2].
Overview of monoclonal antibody biosimilars in Latin America
This article describes the current situation of biosimilar monoclonal antibodies in 10 countries of Latin America and their potential to reduce the cost of antibody therapies.
Challenges with implementing benefit-sharing programmes for biologicals in Europe
Diverse policies have been implemented across Europe to support the rational prescribing of biologicals. Policies limiting the reimbursement of high-cost pharmaceuticals and applying prescription quotas for ‘best-value’ biologicals (BVB) have often been combined with educational campaigns about biosimilars and with benefit-sharing (gainsharing) initiatives [1, 2].
Biocon: cancer biosimilar launch in Canada and US$90 million Malaysia deal
In May 2022, Biocon Biologics launched a bevacizumab biosimilar co-developed with Viatris (formerly Mylan), in Canada; and in April 2022, Biocon was awarded a three-year contract, worth US$90 million, by Malaysia's Ministry of Health
American Academy of Ophthalmology biosimilars position statement published
The American Academy of Ophthalmology issued a policy statement on the use of biosimilars in ophthalmic practice in January 2022 [1].
Canada approves five biosimilars in last seven months
Since November 2021, Canada’s drug regulator, Health Canada, has approved no less than five biosimilars for the treatment of diabetes, cancer, osteoporosis and autoimmune conditions.
Successful increase of biosimilar adoption in a large integrated health delivery network
Biologicals have revolutionized patient care across several therapeutic areas but are associated with significantly higher costs to patients and healthcare systems. Biosimilars offer the opportunity to reduce drug spending without compromising patient care. Medication utilization management (UM), a formulary management methodology, can be used to facilitate the adoption of biosimilars. Author Sophia Humphreys describes an innovative and strategic biosimilar adoption programme that was developed and implemented at Providence St Joseph Health [1]. Providence is a large integrated health system including 53 hospitals, 1,085 clinics and an annual drug spend of >US$1.2 billion.