Biosimilars/Research
Trastuzumab biosimilar Kanjinti remains stable in polyolefin bags and elastomeric devices
A study of the trastuzumab biosimilar ABP 980 (Kanjinti) finds that the compound remains stable in concentrated multi-dose bags and diluted in intravenous (IV) bags and elastomeric devices, providing preparation options to suit different global pharmacy practices.
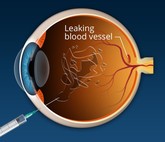
The CROWN study: real-world comparison between ‘similar biologic’ and originator ranibizumab and bevacizumab
The last decade and a half have witnessed a paradigm shift in the way vascular disorders affecting the retina are treated. Anti-vascular endothelial growth factor (VEGF) antibodies have become the mainstay of treatment. These monoclonal antibodies are injected in the vitreous cavity. For the treatment to be effective, repeated injections every 4 to 8 weeks over many years need to be given, which proves to be quite expensive.
Will the unclear path of biosimilar interchangeability become clearer?
Biosimilar use has led to increased access to biological treatments for some molecules wherein switching from originator to biosimilar has been one means to exchange products [1, 2]. Another but less applied way to exchange the products is via substitution by a pharmacist, i.e. without informing the prescriber [2]. Substitution of biosimilars is a topic with many contrasting views and has been highly debated [3-12]. More clarity on the matter is needed because health care has reached a point where policy decisions need to be made about whether substitution should become a practice in biosimilar use. Therefore, researchers from Denmark, Sweden and The Netherlands investigated the views of experts from medicines agencies and the pharmaceutical industry on the science underpinning interchangeability of biosimilars [13].
Europe’s IP framework should support earlier authorization of biosimilars, review finds
A review of the European Pharmaceutical Strategy suggests the framework should focus on intellectual property (IP) initiatives that support the earlier authorization of biosimilars.
Biosimilars and non-medical switching among Saudi rheumatologists. The knowledge gap
The increasing cost of originator biologicals, combined with consequences of the COVID-19 pandemic, have burdened the healthcare system. Unfortunately, the biosimilar uptake in Saudi Arabia has not reached an optimal level despite the approval of several agents. Therefore, physicians are essential stakeholders in the process and acceptance of biosimilars.
The US needs to learn from Europe to increasing access to biosimilars
Europe has been leading the way when it comes to encouraging the use of biosimilars. This is especially true in Scandinavian countries, such as Norway, which are seen as leading the way when it comes to switching and substitution of biosimilars in Europe [1]. In fact, an analysis by Norwegian researchers has found that ‘tremendous savings’ could be realized in the US should the country implement similar strategies [2].
Real-world utilization of adalimumab biosimilar ABP 501
In a poster presentation at the virtual Crohn’s & Colitis Congress, which took place on 20‒22 January 2022, a study describing real-world utilization of adalimumab biosimilar ABP 501 in patients with inflammatory bowel disease (IBD) in Europe was presented [1].
The impact of insulin biosimilars on the diabetes landscape
The cost of diabetes care in the US continues to rise, with insulin accessibility and affordability a particular concern for providers and patients. Increased competition from increased availability of insulin biosimilars may help address these issues.
Differences in immunogenicity, pharmacovigilance and legal documents in biological products in Latin America
A study published in 2021 and carried out by Ramírez-Telles et al. examined the immunogenicity, pharmacovigilance and legal documentation of biological and biotechnological drugs as aspects required in the regulations for the registration of these products. This study was conducted in nine Latin American countries – Brazil, Chile, Costa Rica, Cuba, Dominican Republic, El Salvador, Guatemala, Honduras and Panama – that currently have a regulatory framework for their registration [1].
Biological standardization of bevacizumab: impact on global harmonization of potency assessment
Bevacizumab is a humanized monoclonal antibody (mAb) targeting vascular endothelial growth factor (VEGF), which causes excessive growth of new blood vessels from pre-existing ones in the form of angiogenesis, underlying pathology of cancers and intraocular neovascular diseases. Through binding to soluble VEGF, bevacizumab sterically blocks the interaction of VEGF with VEGF receptor 2 on endothelial cells and thereby interrupts the activation of downstream signalling for angiogenesis. Bevacizumab is indicated for the treatment of a range of cancers, including metastatic colorectal cancer, metastatic breast cancer and non-small-cell lung cancer. It is also used for off-label treatment of eye diseases including neovascular age-related macular degeneration and diabetic macular oedema.