Biosimilars/News
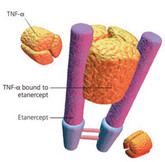
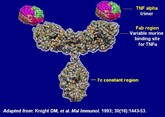
Coherus starts phase III biosimilar etanercept trial
US-based biosimilars developer Coherus BioSciences (Coherus) announced on 23 June 2014 the start of a global phase III trial for a biosimilar version of etanercept.
Biosimilar infliximab receives approval in Japan and Turkey
South Korean biotechnology company Celltrion announced on 4 and 16 July 2014 that the company had received marketing approval for its biosimilar Remsima (infliximab) in Japan and Turkey, respectively.
FDA accepts biosimilar filgrastim application
Sandoz, the generic drug division of Swiss drug giant Novartis, announced on 24 July 2014 that the US Food and Drug Administration (FDA) had accepted its application for approval of the company’s biosimilar filgrastim product.
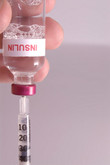
EMA approves biosimilar insulin
The European Medicines Agency’s (EMA’s) Committee for Medicinal Products for Human Use (CHMP) announced on 27 June 2014 that it had recommended granting of marketing authorization for a biosimilar insulin glargine product (LY2963016).
Oncobiologics and IPCA create biosimilars alliance
US-based Oncobiologics announced on 2 June 2014 the creation of a two-part alliance for the development, manufacture and commercialization of biosimilar monoclonal antibodies with Indian generics maker IPCA Labs (IPCA).
Biosimilars developer Coherus raises US$55 million investment
US-based biosimilars developer Coherus BioSciences (Coherus) announced on 19 May 2014 that it had raised investments of US$55 million. New investors KKR & Co LP, Venrock, RA Capital Management, Rock Springs Capital and Fidelity Biosciences joined existing investors Sofinnova Ventures, Lilly Ventures and Vivo Capital.
Celltrion starts phase III biosimilar trastuzumab trial
South Korean biotechnology company Celltrion is starting a phase III clinical trial for its biosimilar trastuzumab candidate (CT-P6) in patients with human epidermal growth factor receptor 2-positive (HER2+) early breast cancer, according to the European Union’s Clinical Trials Register.
Alvotech and Finesse enter into biosimilars collaboration
Iceland-based biopharmaceutical company Alvotech and US-based bio-process management company Finesse Solutions (Finesse) announced on 14 March 2014 that they had entered into a partnership for bio-manufacturing.
Biosimilar filgrastim gains Japanese approval
Sandoz (the generics unit of Novartis) announced on 24 March 2014 that its subsidiary Sandoz Japan had received marketing authorization approval for its biosimilar filgrastim (Filgrastim BS Sandoz).
Long-acting G-CSF lipegfilgrastim launched in UK
Teva UK, a subsidiary of generics giant Teva Pharmaceutical Industries (Teva) announced on 24 February 2014 the launch of its long-acting granulocyte colony-stimulating factor (G‑CSF) Lonquex (lipegfilgrastim) in the UK.