Biosimilars
EMA recommends approval of first ustekinumab biosimilar Uzpruvo
On 9 November 2023, the European Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion, recommending the granting of a marketing authorisation for the medicinal product Uzpruvo (ustekinumab), a biosimilar of reference product Stelara, intended for the treatment plaque psoriasis, including paediatric plaque psoriasis, psoriatic arthritis and Crohn’s disease in adults.
FDA approves first interchangeable ustekinumab biosimilar Wezlana
On 31 October 2023, the US Food and Drug Administration (FDA) approved Wezlana (ustekinumab-auub) as a biosimilar to and interchangeable with Stelara (ustekinumab) for multiple inflammatory diseases.
Canada’s Prince Edward Island adopts biosimilars switching policy
Canada’s Prince Edward Island (PEI) becomes the 11th Canadian jurisdiction to announce the adoption of biosimilars switching policies.
Impact of trastuzumab biosimilars use in metastatic HER2-positive breast cancer
A review conducted by Sarder and Ahmad attempted to describe and analyse the studies published in peer-reviewed literature on the use of trastuzumab biosimilars for patients with HER2-positive breast cancer, including their efficacy, safety, pharmacokinetic and pharmacodynamic properties [1].
Alvotech biosimilars: FDA ustekinumab application rejection; adalimumab interchangeability designation re-submission
In October 2023, Iceland-based biosimilar manufacturer Alvotech, announced that the US Food and Drug Administration (FDA) has issued a complete response letter (CRL) for Alvotech’s Biologics License Application (BLA) for AVT04, a biosimilar candidate to Stelara (ustekinumab). As such, the application was not approved. This follows Alvotech’s September 2023 resubmission of a BLA for their high-concentration biosimilar formulation to AbbVie’s blockbuster arthritis treatment Humira (adalimumab), AVT02 [1], following receipt of a similar CRL.
Japanese approval for first ustekinumab biosimilar
Iceland-based biosimilar manufacturer Alvotech announced on 25 September 2023 that its commercialization partner in Japan, Fuji Pharma Co Ltd (Fuji) had received marketing approval from Japan’s medicines regulatory agency, the Pharmaceuticals and Medical Devices Agency (PMDA), for its ustekinumab biosimilar AVT04 in Japan.
ASCO policy statement on biosimilars and interchangeables in oncology updated
The American Society of Clinical Oncology (ASCO) has released a new policy statement on the use of biosimilar and interchangeable products in oncology.
The statement proposes guidance and recommendations for stakeholders, including manufacturers, payers, regulatory agencies, clinicians, and patients, to ensure equitable access to high-quality care and to address challenges to biosimilar uptake [1].
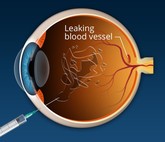
Biosimilar anti-VEGF: transforming retina treatment economics in South Asia
Anti-vascular endothelial growth factors (anti-VEGF) agents combat retinal diseases causing blindness. However, high costs and non-adherence pose challenges. Ophthalmic anti-VEGF ranibizumab biosimilars, which have received multiple approvals in South Asia, are helping to reduce healthcare costs and revolutionize ophthalmic treatments.
Long-term data support the clinical comparability of AVT04 to Stelara
Long-term efficacy, safety and tolerability data for the AVT04 ustekinumab biosimilar candidate developed in partnership by Alvotech and STADA corroborate the finding of therapeutic equivalence to the Stelara® reference product in patients with moderate-to-severe chronic plaque psoriasis (PsO) in the primary endpoint at Week 12.
FDA approves first tocilizumab biosimilar Tofidence
On 29 September 2023, the US Food and Drug Administration approved Tofidence (tocilizumab-bavi) as the first biosimilar to Actemra (tocilizumab). Tofidence is first-of-its-kind biosimilar tocilizumab to receive approval in the US.